Varicose Veins

Varicose Veins

- VVT Medical
- >>
- Varicose Vein
Overview
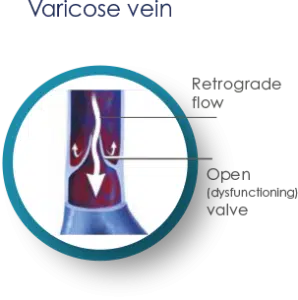
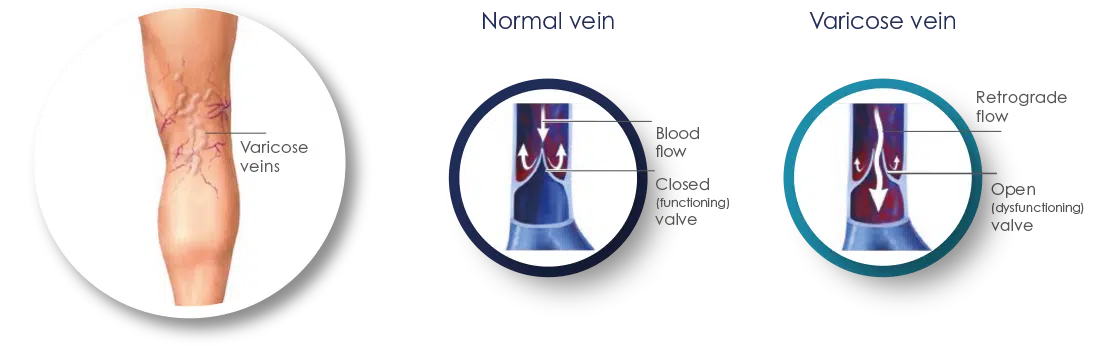
Varicose veins are enlarged, swollen and twisted veins, appearing blue or dark purple in color. They result from a disorder in the circulatory system where faulty valves in the veins allow blood to flow in the wrong direction or to “pool.”

More than 70 million people across the United States and Europe suffer from varicose veins. This includes approximately 23% of adults in the U.S. and 50% of adults over 50. Women are more prone to developing varicose veins and make up 60% of those affected. Risk factors include pregnancy, family history and obesity.
Since the GSV is primarily responsible for returning blood to the heart via the femoral vein, it is considered the main underlying cause of varicose veins and is the most commonly treated.
When veins become varicose, the valves in the saphenous veins in our legs no longer close properly, resulting in Venous Valve Incompetence. This causes the blood to accumulate in the lower leg instead of flowing upward to the heart, through the lungs, replenishing it with oxygen and circulating throughout the body. This “pooling” of the blood in the lower legs, also known as Reflux Disease, is a chronic condition that leads to enlarged veins and corresponding medical and cosmetic issues.
Chronic leg ulcers can often take five or six weeks to heal. The most frequent cause of such ulcers is a disease in the leg veins. In fact, venous disease is responsible for about 80% of cases. Ulcers are extremely difficult to cure without treating the affected vein; serious complications can follow a long period of non-treatment.
Symptoms & Diagnosis

- Leg pain, fatigue & heaviness
- Swollen ankles and calves
- Restless Leg Syndrome
- Skin discoloration, matting, induration
- Visible tortuous varicosities
- Pigmentation, eczema, lipodermatosclerosis
- Skin ulcers- incurable wounds
- Burning and itching skin
- Skin sensitivity, susceptibility to outside impact, hemorrhage, thrombophlebitis.
- Blood clots
- Increased risk for DVT and tissue necrosis